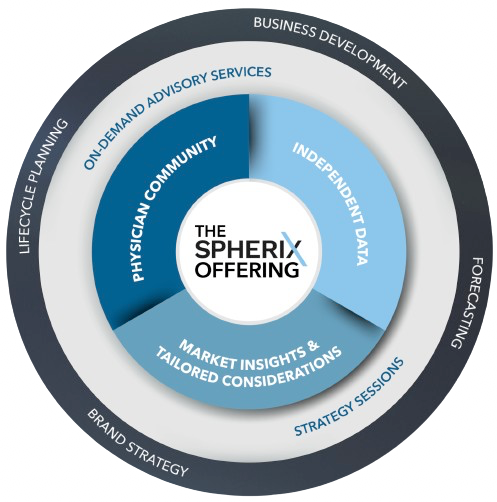
Perspective
is everything.
Across the pharma lifecycle, an accurate
and complete market view is essential to
stay ahead of the curve and uncover
emerging healthcare trends.
and complete market view is essential to
stay ahead of the curve and uncover
emerging healthcare trends.